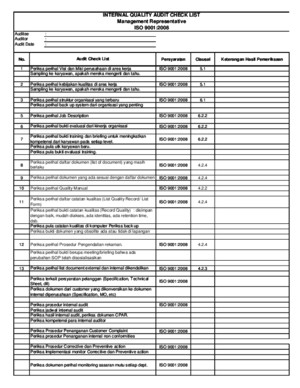
Internal Quality Audit Checklist ISO 13485:2003 ISO 9001:2000
There is document - Internal Quality Audit Checklist ISO 13485:2003 ISO 9001:2000 available here for reading and downloading. Use the download button below or simple online reader.
The file extension - PDF and ranks to the Instruction manuals category.
Tags
Related
Comments
Log in to leave a message!
Description
Quality Audit checklist for Medical Device quality audits This checklist is set up to evaluate a quality system against two possible specifications, ISO 9001:2000 or ISO 13485:2003 Check the appropriate box under Quality System on page 1 If the quality system being evaluated is not designed to be compliant with one of these specifications, check Other, note the specification it is compliant to, and use the checklist for ISO 9001:2000
Transcripts
Recommended