Chemistry SPM Paper 3 Question Analysis From 2003
There is document - Chemistry SPM Paper 3 Question Analysis From 2003 available here for reading and downloading. Use the download button below or simple online reader.
The file extension - PDF and ranks to the Documents category.
Tags
Related
Comments
Log in to leave a message!
Description
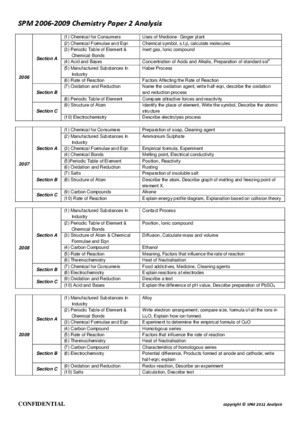
Chemistry SPM Paper 3 Question Analysis from 2003 ² 2009 Year 2009 Structure Question 1 Effect of type of electrode on the electrolysis of aqueous solution 2 Rate of reaction 1 Effect of ethanoic acid and ammonia solution on the coaagulation of latex 1 Exothermic & endothermic reaction Planning an Experiment 1 Relationship between concentration of nitric acid & pH value 1 Reactivity of lithium, sodium and potassium with water 2008 2007 2006 2005 2004 2003 1 Construct electrochemical serie
Transcripts
Chemistry SPM Paper 3 Question Analysis from 2003 ² 2009Year Structure Question Planning an Experiment 2009 1 Effect of type of electrode on theelectrolysis of aqueous solution2 Rate of reaction1 Relationship betweenconcentration of nitric acid pHvalue2008 1 Effect of ethanoic acid andammonia solution on thecoaagulation of latex1 Reactivity of lithium, sodiumand potassium with water2007 1 Exothermic endothermicreaction1 Construct electrochemicalseries to determine factor thatinfluence the difference in voltage2006 1 Heat of neutralization 1 Compare iron steel from theaspect of hardness/resistant tocorrosion2005 1 Freezing point of naphthalene2 Electrochemical series based onpotential difference1 Effect of alloying on thehardness of metal2 How concentration affect therate of reaction of acid with metal2004 1 Empirical formula of magnesium oxide2 Reactivity series of metals1 Prepare esters from twodifferent alcohol2003 1 Effect of temperature on therate of reaction2 Construct ionic equationsthrough continuous variationmethod1 Compare the elasticity of vulcanized rubber unvulcanized rubber Common Chemicals Common salt - sodium chloride Table Sugar - sucroseVinegar - ethanoic acidVitamin C - ascorbic acidBaking soda - sodium bicarbonateDry ice - solid carbon dioxideAluminium foil - aluminiumCopper wire - copperClean sand - silicon dioxideDiamond - carbon crystal
Recommended